Pcl3 electron domain geometry
When we talk about the hybridization of PCl 3 students should not confuse it with PCl5.
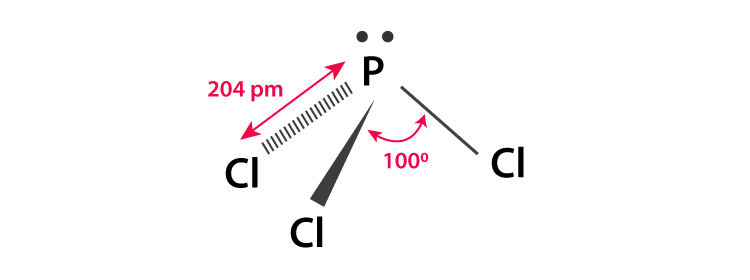
The Lewis structure of PCl3 consists of a central phosphorus atom P and three external chlorine atoms Cl. There are three single bonds between the phosphorus atom P and each of the chlorine atoms Cl. There is one lone pair of electrons on the phosphorus atom P and three lone pairs of electrons on each of the chlorine atoms Cl. The PCl3 Lewis structure is shown below:. Phosphorus and chlorine are elements of group 15 and 17 of the periodic table, respectively. The central atom must be highly or minimally electronegative. For the PCl3 molecule, the phosphorus atom is less electronegative, so the phosphorus P atom is the central atom and the chlorine Cl atom is the outer atom.
Pcl3 electron domain geometry
.
Looking at the PCl 3 molecular geometry it is trigonal pyramidal with a bond angle of approx. The central atom must be highly or minimally electronegative.
.
We begin by assuming a Lewis structure model for chemical bonding based on valence shell electron pair sharing and the octet rule. We thus assume the nuclear structure of the atom, and we further assume the existence of a valence shell of electrons in each atom which dominates the chemical behavior of that atom. A covalent chemical bond is formed when the two bonded atoms share a pair of valence shell electrons between them. We know that double bonds are generally stronger and have shorter lengths than single bonds, and triple bonds are stronger and shorter than double bonds. We should expect that the properties of molecules, and correspondingly the substances which they comprise, should depend on the details of the structure and bonding in these molecules. The relationship between bonding, structure, and properties is comparatively simple in diatomic molecules, which contain two atoms only, e. A polyatomic molecule contains more than two atoms. An example of the complexities which arise with polyatomic molecules is molecular geometry: how are the atoms in the molecule arranged with respect to one another? In a diatomic molecule, only a single molecular geometry is possible since the two atoms must lie on a line. However, with a triatomic molecule three atoms , there are two possible geometries: the atoms may lie on a line, producing a linear molecule, or not, producing a bent molecule.
Pcl3 electron domain geometry
We continue our discussion of structure and bonding by introducing the valence-shell electron-pair repulsion VSEPR model A model used to predict the shapes of many molecules and polyatomic ions, based on the idea that the lowest-energy arrangement for a compound is the one in which its electron pairs bonding and nonbonding are as far apart as possible. Keep in mind, however, that the VSEPR model, like any model, is a limited representation of reality; the model provides no information about bond lengths or the presence of multiple bonds. Although the VSEPR model is a simple and useful method for qualitatively predicting the structures of a wide range of compounds, it is not infallible. In this section we will make the connection between hybrid orbital described in Chapter 6. The hybrid orbital picture, although more complex, provides a better explanation of such things. Instead, many of these species, including SrF 2 and BaF 2 , are significantly bent. A more sophisticated treatment of bonding is needed for systems such as these. The VSEPR model can predict the structure of nearly any molecule or polyatomic ion in which the central atom is a nonmetal, as well as the structures of many molecules and polyatomic ions with a central metal atom. Instead, it is a counting procedure that accurately predicts the three-dimensional structures of a large number of compounds, which cannot be predicted using the Lewis electron-pair approach. Lewis electron structures predict the number and types of bonds, whereas VSEPR can predict the shapes of many molecules and polyatomic ions.
Jeongyeon legs
The central atom must be highly or minimally electronegative. Therefore, the Lewis structure of PCl3 in the above step is stable and has not changed further. The P-Cl bonds are basically formed when the sp 3 hybrid orbitals overlap with 3p orbitals of chlorine which are singly occupied. The valence shell in phosphorus is 5. Did not receive OTP? You may like Is sodium a metal or nonmetal? All in all, if we look at PCl3, there are 3 electron pairs, 3 bond pairs and one lone pair of electrons. Step 4 Stability of structure In step 3, for the PCl3 molecule, we can see that there are three lone pairs of electrons on each of the outer chlorine atoms, forming an octet, so they are stable. Download Now. The PCl3 Lewis structure is shown below: Steps for drawing the PCl3 Lewis structure Step 1 Calculate the number of valence electrons for P and Cl Phosphorus and chlorine are elements of group 15 and 17 of the periodic table, respectively. Phosphorus in PCl3 undergoes sp3 hybridization, involving the combination of one s orbital and three p orbitals of phosphorus to form four sp3 hybrid orbitals. Does it have to be refrigerated? Step 2 Identify the central atom The central atom must be highly or minimally electronegative. Watch Now. Phosphorus trichloride
Phosphorus trichloride with a chemical formula PCl3 is a yellow fuming liquid. This liquid can be colorless as well. PCl3 is a toxic liquid with an unpleasant smell.
Post My Comment. These hybrid orbitals overlap with the p orbitals of chlorine atoms, creating sigma bonds and maintaining the trigonal pyramidal molecular shape of PCl3. Share Share Share Call Us. The molecular shape of PCl3 is trigonal pyramidal. There are two single bonds between the xenon atom Xe and each fluorine atom F The bond angles between the central phosphorus atom and the three chlorine atoms are approximately PCl 3 is sp 3 hybridized. There are three single bonds between the phosphorus atom P and each of the chlorine atoms Cl. The PCl3 Lewis structure is shown below:. Does it have to be refrigerated?

You are not right. Let's discuss.
Thanks for the help in this question, the easier, the better �