The ionization energy of hydrogen atom is 13.6
Get Started.
Isotopes: The elements which have the same atomic number but different mass numbers are called isotopes. Last updated on May 25, Get Started. SSC Exams. Banking Exams. Teaching Exams.
The ionization energy of hydrogen atom is 13.6
The ionisation potential of hydrogen atom is The ionization potential of hydrogen atom is When an electron in the hydrogen atom in ground state absorb a photon of energy If ioinsation potential of hydrogen atom is The ionization energy of hydrogen atom is Hydrogen atoms in the ground state are excited by electromagnetic radiation of energy How many spectral lines will be emitted by the hydrogen atoms. What will happen if a hydrogen atom absorbs a photon of energy greater than Ionisation potential of hydrogen atom is Hydrogen atom in ground state is excited by monochromatic light of energy The spectral lines emitted by hydrogen according to Bohr's theory will be. The ionisation potential of H-atom is The energy required to remove an electron from the second orbit of hydrogen is. The ionisation potential of ground state of hydrogen atom is
India post Postman. Krushi Vibhag Maharashtra Senior Clerk.
Ionization potential of hydrogen atom is Hydrogen atoms in the ground state are excited by monochromatic radiation of photon energy The spectral lines emitted by hydrogen atoms according to Bohr's theory will be. The ionization potential of H-atom is The H-atoms in ground state are excited by mono chromatic radiations of photon energy Then the number of spectral lines emitted by the excited atoms, will be. The ionization energy of hydrogen atom is
Last time, we discussed electromagnetic waves and how light is quantized as photons. We related the energy of a photon to its frequency and wavelength using the Planck-Einstein relation:. Electron volts are also a unit of energy, but they are much smaller than a joule. We can convert between joules and electron volts using. Either one works - double checking how your units work out is a great way to check your answer. Planck's constant is. If a photon with sufficient energy is absorbed by an atom, the atom can become ionized : it loses an electron! The electron is then ejected from the atom, and flies through space. Determine a the minimum energy state the electron could have been in, and how much energy would have been leftover, b the velocity of the electron right when it leaves. The energy to ionize from the 8th energy level is.
The ionization energy of hydrogen atom is 13.6
In physics and chemistry , ionization energy IE is the minimum energy required to remove the most loosely bound electron of an isolated gaseous atom , positive ion , or molecule. Roughly speaking, the closer the outermost electrons are to the nucleus of the atom , the higher the atom's ionization energy. In physics, ionization energy is usually expressed in electronvolts eV or joules J.
Invoke-azrestmethod
Indian Navy Agniveer. Bihar Vidhan Sabha Office Attendant. Bihar Vidhan Parishad Security Guard. UP Police Constable. CG Lab Attendant. MBA Entrance Exam. A charge Q is situated at the corner of a cube the electric flux passe JK Police Constable. Karnataka Bank PO. Deuterium: It is the isotope of hydrogen which has atomic number 1 and mass number 2.
The energies of electrons in molecular orbitals can be observed directly by measuring the ionization energy. This is the energy required to remove an electron, in this case, from a molecule:. Therefore, it requires more energy to remove an electron from the hydrogen molecule than from the hydrogen atom; the electron therefore has a lower energy in the molecule.
Karnataka TET. Telangana High Court Office Subordinate. Gujarat TAT. Rajasthan Fireman. BSF Constable. Indian Navy Chargeman. NVS Staff Nurse. Railway TTE. Important Exams. WB Police Constable. AP TET. Civil Services Exam. FCI Manager.

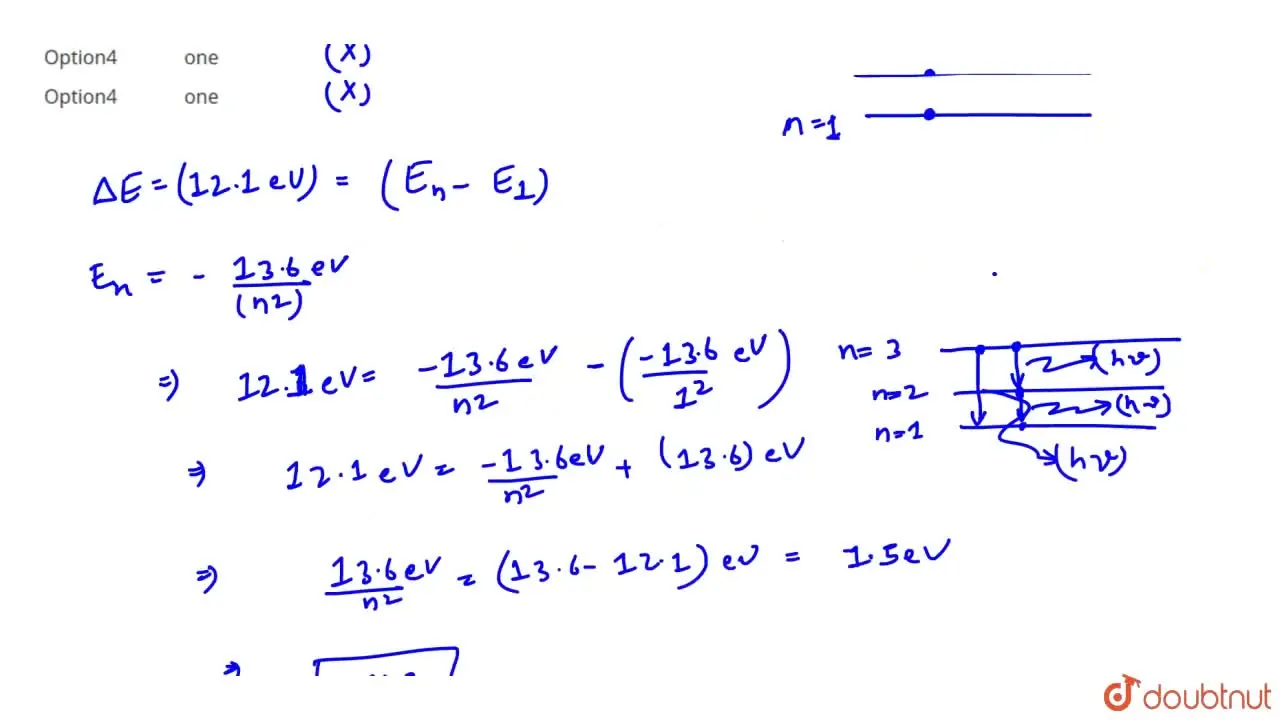
0 thoughts on “The ionization energy of hydrogen atom is 13.6”