Arrange the following in the increasing order of boiling point
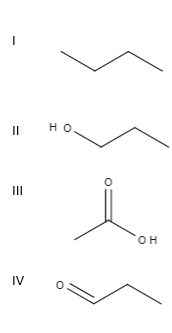
Alcohol has the highest boiling point due to more extensive intermolecular H-bonding. Aldehyde is more polar than ether so, ethanal has high BP than ethyl ether. And alkane has the lowest BP. Dont't have an account?
Arrange the following : In increasing order of basic strength : a Arrange the following : In decreasing order of basic strength in gas Arrange the following : In increasing order of boiling point : C Arrange the following : In increasing order of solubility in water How will you convert : Ethanoic acid into methanamine.
Arrange the following in the increasing order of boiling point
Do you know how to arrange bromomethane chloromethane and chloroform in order of increasing boiling point? Learn the concept of intermolecular forces and their effect on boiling point with Toppr the best online learning platform for students. Ask any Organic Chemistry question and get answers from experts. Continue reading Arrange the compounds in order of decreasing boiling point. Arrange the following compounds in order of increasing boiling point. Arrange the following compounds in order of properties a Pentanol Pentanol ethanol Propanol and methanol. Problem 6. Neon and HF have approximately the same molecular masses. Explain why neon and HF have different boiling points.
Com Master of Commerce M.
Do you know how bromomethane bromoform chloromethane and chloroform are arranged in order of increasing boiling point? Learn the concept of intermolecular forces and their effect on boiling point with Toppr, the best online learning platform for students. Ask any organic chemistry question and get expert answers. Continue reading Arrange the compounds in decreasing order of boiling point. Arrange the following compounds in increasing order of their boiling point.
Arrange each of the following sets of compounds in order of increasing boiling point temperature:. Download Guided Solution as a pdf. The stronger the intermolecular forces, the more energy required to break the intermolecular forces and transition the compound between the liquid phase and the gas phase. All forces are the same type therefore it goes based on size. The largest atom has the highest boiling point temperature. All are nonpolar with dispersion forces. All molecules have the same elements but differ in the numbers of C and H and the molecule size. The larger the molecule larger size, molar mass and surface area , the higher the boiling point temperature. The question asked you to arrange in order of increasing boiling point temperature, so the lowest boiling point is first. The molecules will be arranged in order of weakest intermolecular attractions to the strongest going from left to right.
Arrange the following in the increasing order of boiling point
Arrange the following sets of compounds in order of their increasing boiling points: A : Pentanol, butanol, butanol, ethanol, propanol, methanol B : Pentanol, n-butane, pentanal, ethoxyethane. Number of carbon atoms: As the number of carbon atoms increases in alcohol molecule, the boiling point of alcohol also increases as the molecular mass increases which increases the molecular attraction between molecules. Branching in molecules: As the number of branching increases, the boiling point of alcohol decreases because branching in molecules decreases the surface area thereby decreasing the attractive forces between individual molecules. Hydrogen bonding: As the extent of hydrogen bonding increases, the boiling point also increases due to increase in the attractive forces between individual molecules. Moreover, dipole-dipole interactions are also similar in both the compounds only hydrogen bonding is making a difference as extent of intermolecular hydrogen bonding is greater in case of pentanol than pentanal. Hence, boiling point of pentanol is greater than pentanal. Arrange the following compounds in increasing order of boiling point. Propan - 1 - ol, butan - 1 - ol, butan - 2 - ol, pentan - 1 - ol a Propanol, butanol, butan - 1 - ol, pentano b Propanol, butan - 1 - ol, butanol, pentanol c Propanol, butanol, butanol, pentanol d Propanol, butanol, butanol, pentanol. Byju's Answer. Open in App.
36ff
Arrange the following : In decreasing order of basic strength in gas Was this answer helpful? How will you convert : Propanoic acid into ethanoic acid? Go To Your Study Dashboard. Solved Arrange these compounds in increasing order Arrange each compound in order of increasing boiling point Accomplish the following conversions : Chlorobenzene to p-chloranili Arrange the following compounds in descending order Learn Change. Latest Question A sum of money under compound interest doubles itself in 4 years. Problem 6. Pharma M.
As you know, a molecule's boiling point depends on the strength of the intermolecular forces of attraction its molecules exhibit. In your case, you have to find how the boling points of three nonpolar molecules relate to each other. The halogens, which are the lements that make up group 17 of the periodic table , exist as diatomic molecules.
In how many years will it become 16 times itself? Study Abroad Change. Your Answer. Do you know how to arrange bromomethane chloromethane and chloroform in order of increasing boiling point? Arrange the following compounds in order of properties a Pentanol Pentanol ethanol Propanol and methanol. How will you convert : Nitromethane into dimethylamine. How will you convert : Hexanenitrile into 1-aminopentane Text Solution. Hospitality and Tourism Change. Competition Change. Arrange the following compounds in the order of properties indicated a Pentanol Pentanol Ethanol Propanol and Methanol. Arrange the following compounds in increasing order of Latest Question A sum of money under compound interest doubles itself in 4 years. How will you convert : Methanol to ethanoic acid. Increasing order in their boiling points- Alcohol has the highest boiling point due to more extensive intermolecular H-bonding.

I will know, I thank for the information.
The intelligible answer